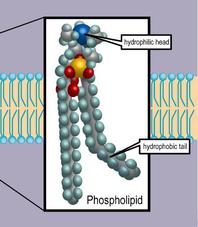
Saturated fatty acid tails only have single bonds, so every carbon atom is bonded to four other atoms. In this context, saturation refers to the number of single bonds a carbon atom has with other molecules. An important property to keep in mind of these lipids is their degree of saturation. They contain a hydrophilic, polar phosphate head group joined through an ester linkage to a hydrophobic nonpolar fatty acid tail. Phospholipids, or phosphatides, are the primary component of the phospholipid bilayer of the cell membrane. It’s important not to get these terms confused!įor the MCAT, the two main structural lipids you should be familiar with are phospholipids and sphingolipids. For instance, an amphoteric compound can react as an acid or a base, and an amphiprotic substance can donate or receive a proton. You may recall similar-sounding words with quite different meanings. You can find more information about steroid hormones in our guide on the endocrine system. Steroid hormones are specially secreted by endocrine glands and act as hormones. It plays an integral role in our cell membranes (more on this later) and is a precursor for many molecules, including steroid hormones. The most commonly tested steroid on the MCAT, cholesterol, is one you should be very familiar with. As you’d expect, these hydrocarbon rings make steroids nonpolar.

Steroids are characterized by their four-ring structure, which includes three cyclohexanes and one cyclopentane. These signaling lipids are further divided into two categories: steroids and fat-soluble vitamins. Signaling lipids are specialized lipids involved in signal transduction pathways, the passing of information between and within cells. The structure of waxes is characterized by long, alkane chains. Waxes are protective secretions that serve as a waterproofing compound for many plants and animals. All cell membranes are composed of lipids (as we will discuss), and many animals use lipids as a form of energy storage. This is a unifying feature of all lipids and has special implications for how lipids are used in the body.ĭue to their nonpolar properties, lipids have assumed many different functions within organisms. The hydrocarbon ring structure of testosterone and the hydrocarbon tail of the triglyceride will cause the molecules to be nonpolar and insoluble in polar solvents (such as water). At the end, we’ll include some practice passages and discrete questions so you can apply your knowledge in the exact context the AAMC will on test day.Īlthough they may look very different, they are both hydrophobic lipids.
In this guide, we will explain everything you need to know about lipids to be successful on the MCAT. Indeed, they are a likely topic to show up on your exam. Lipids are everywhere in your body-from hormones, cellular structures, and more. The diversity of roles that lipids hold often makes them a challenging topic for students during their MCAT prep. Minimizing carbohydrates while consuming more fat seemed to alleviate seizures while still providing children the nutrients they needed to properly develop.įats, also known as lipids, are relatively simple molecules responsible for a variety of functions in our body, including energy storage and transmitting signals. In the early twentieth century, one of the most common therapies to control seizures in pediatric patients suffering from epilepsy was not a medication but a diet.

